Checkmate therapeutics8/11/2023  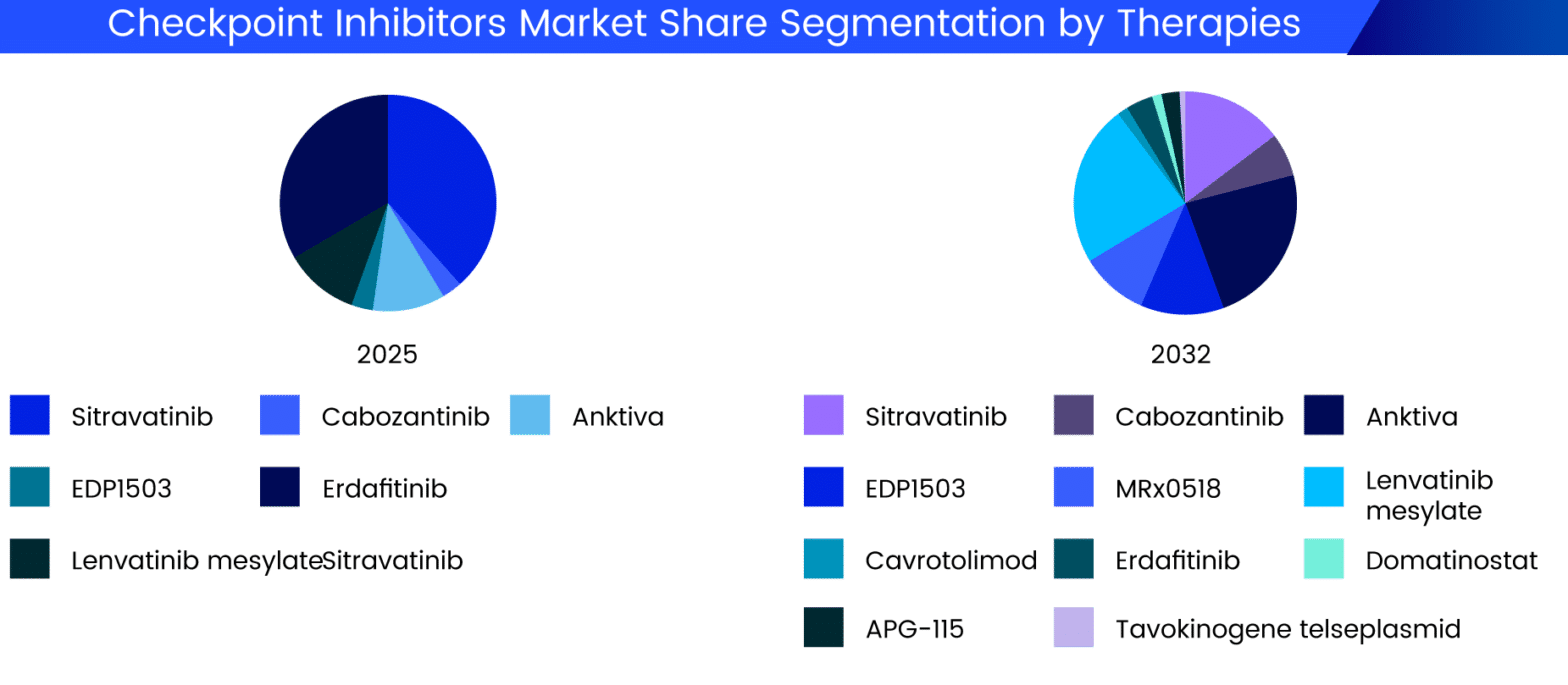
In the filing, it specified that it currently had 50,000 doses and that it expected to reach a total of 300,000 doses "within the next few months." The FDA granted approval for emergency use authorization in November 2020. On October 7, Trump posted a five-minute video to Twitter reasserting that this drug should be "free." That same day, Regeneron filed with the FDA for emergency use authorization. His doctors obtained it from Regeneron via a compassionate use request (as clinical trials had not yet been completed and the drug had not yet been approved by the US Food and Drug Administration (FDA)). President Donald Trump was infected with COVID-19 and taken to Walter Reed National Military Medical Center in Bethesda, Maryland, he was administered REGN-COV2. 
California based laboratory, FOMAT, is part of the clinical investigation through their doctors Augusto and Nicholas Focil. Regeneron similarly said in its own news release that same day that "the government has committed to making doses from these lots available to the American people at no cost and would be responsible for their distribution," noting that this depended on the government granting emergency use authorization or product approval. "By funding this manufacturing effort, the federal government will own the doses expected to result from the demonstration project," the government said in its July 7 news release. 
The $450 million came from the Biomedical Advanced Research and Development Authority (BARDA), the DoD Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense, and Army Contracting Command. In July 2020, under Operation Warp Speed, Regeneron was awarded a $450 million government contract to manufacture and supply its experimental treatment REGN-COV2, an artificial "antibody cocktail" which was then undergoing clinical trials for its potential both to treat people with COVID-19 and to prevent SARS-CoV-2 coronavirus infection. Department of Health and Human Services, which already worked with Regeneron, announced that Regeneron would pursue monoclonal antibodies to fight COVID-19. In April 2022, the business announced it would acquire Checkmate Pharmaceuticals for around $250 million, enhancing its number of immuno-oncology drugs. Prior to the transaction, Sanofi held 23.2 million Regeneron shares. 19.2 million of its shares for around $5 billion, held directly by Sanofi. In May 2020, Regeneron announced it would repurchase approx. Such deals are not unusual for routine drug development in the American pharmaceutical market. This deal was criticized in The New York Times. government would fund 80% of the costs for Regeneron to develop and manufacture antibody-based medications, which subsequently, in 2020, included their COVID-19 treatments, and Regeneron would retain the right to set prices and control production. In October 2017, Regeneron made a deal with the Biomedical Advanced Research and Development Authority (BARDA) that the U.S. Regeneron Pharmaceuticals was home to the two highest-paid pharmaceutical executives as of 2020. In 2019, Regeneron Pharmaceuticals was announced the 7th best stock of the 2010s, with a total return of 1,457%. In July 2015, the company announced a new global collaboration with Sanofi to discover, develop, and commercialize new immuno-oncology drugs, which could generate more than $2 billion for Regeneron, with $640 million upfront, $750 million for proof-of-concept data, and $650 million from the development of REGN2810. The new drug would target the PCSK9 gene. On March 26, 2012, Bloomberg announced that Sanofi and Regeneron were in development of a new drug that would help reduce cholesterol up to 72% more than its competitors. VEGF is a protein that normally stimulates the growth of blood vessels, and interleukin-1 is a protein that is normally involved in inflammation. Regeneron has developed aflibercept, a VEGF inhibitor, and rilonacept, an interleukin-1 blocker. The company was founded by CEO Leonard Schleifer and scientist George Yancopoulos in 1988. Originally focused on neurotrophic factors and their regenerative capabilities, giving rise to its name, the company then branched out into the study of both cytokine and tyrosine kinase receptors. is an American biotechnology company headquartered in Westchester County, New York.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
